New surgical ‘iKnife’ can tell cancerous from healthy tissue
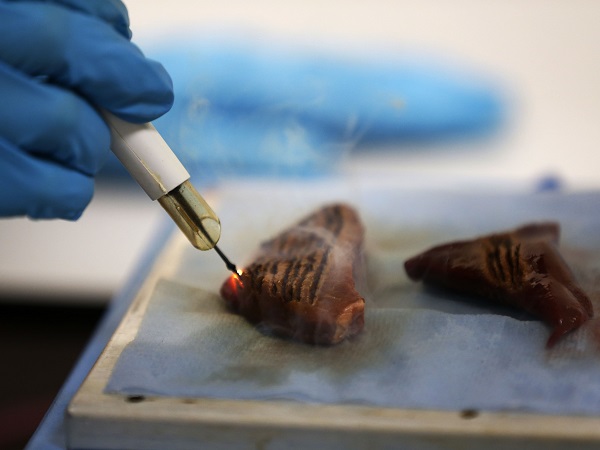
A member of ‘intelligent knife’ development team uses the knife on a piece of animal muscle during a demonstration at St Mary’s Hospital in London, Wednesday, July 17, 2013. Surgeons may have a new way to smoke out cancer.An experimental surgical knife can help surgeons make sure they’ve removed all the cancerous tissue, doctors reported Wednesday. Surgeons typically use knives that vaporize tumors as they cut, producing a sharp-smelling smoke. The new knife analyzes the smoke and can instantly signal whether the tissue is cancerous or healthy. AP/Sang Tan
WASHINGTON – A new kind of surgical knife can tell cancerous from healthy tissue in seconds and may help improve tumor removal in the operating room, according to research published Wednesday.
Known as the iKnife, the tool analyzes the vapor given off as surgeons use electrical current to cut away tissue — and it reports in real time whether the tissue is cancerous or not.
Tests in 91 human patients have shown the “tool’s diagnoses were extremely accurate,” and may be “reliable enough to begin widespread use in operating rooms,” said the study in the US journal Science Translational Medicine.
The iKnife uses mass spectrometry to examine the surgical smoke given off by evaporating tissue, alerting the surgeon in three seconds as to what it contains.
Other current techniques — which include sending removed tissue to a pathology lab for analysis — are “costly…frequently inadequate,” and take about 20-30 minutes, said the researchers from Hungary and Britain.
“Remarkably, there are almost no technologies in routine clinical practice to assist the surgeon in improving the accuracy of cancer tissue clearance,” said the study.
For patients, this uncertainty can lead to repeat surgeries or worse, since returning to the operating table is not always an option for soft tissue tumors, the researchers said.
The iKnife “can augment current tumor diagnostics, and it has the potential to influence ‘on-table’ decision-making and ultimately to improve oncological outcomes,” said the study.
In Britain alone, there are 300,000 new cases of cancer each year and 1.8 million surgical procedures performed in treatment, according to background information in the article.
The iKnife was tested on tissue samples from 302 patients, including those with tumors of the stomach, colon, liver, lung, breast and brain, and later in the operating room during 91 procedures to cut tumor tissue away.
The iKnife, known formally as rapid evaporative ionization mass spectrometry (REIMS), matched post-operative analysis by histopathology in all 91 cases, the study said.
The iKnife is not yet commercially available and likely will not be for at least another year, co-author Jeremy Nicholson of Imperial College London told AFP.
“The machines are modified from existing commercial mass spectrometers,” he said in an email.
“The interface between the knife and the mass spectrometer contains some critical modifications that make it work robustly,” he said, adding that the research machines currently “cost a few hundred thousand pounds.”
According to Stephanie Bernik, chief of surgical oncology at Lenox Hill Hospital in New York, the study is “impressive.”
“If perfected, this would help surgeons and patients avoid countless trips back to the operating room to try and obtain clear margins,” said Bernik, who was not involved with the research.
“A larger study needs to be carried out, but this preliminary data is very exciting,” she said.
For Richard Fogler, chairman emeritus of the department of surgery at Brookdale University Hospital and Medical Center in Brooklyn, New York, the technique might save some time, and it could be of particular use for plastic surgeons and for removal of head and neck cancers.
However, he said it would take a lot more study to change the standard of care in tumor removal, which calls for a certain margin around cancer tissue to be taken out depending on the severity of the cancer.
“It sounds like ‘we discovered a magic wand,'” said Fogler, who took issue with the paper’s claim that the iKnife had the potential “to improve oncological outcomes.”
“I think it has been pretty well shown over the years you can’t make a statement about oncologic outcome without knowing what the stage of the disease is,” Fogler said.
“It doesn’t matter how you take it out. It doesn’t matter what you use to biopsy it.”